Understanding Radiation: Half Life

Related Articles
Following the recent problems at the Fukushima No.1 Nuclear Power Plant, the news has been teeming with talk of isotopes, radiation, contamination, and health risks. For many of us, all this talk of half lives and microSieverts can be a little confusing. In order to understand what we are being told in the news, we need to know a little about nuclear physics. The following series of articles will introduce a few fundamental concepts to help us better understand recent events (don’t worry, we’re going to keep this simple).
In a previous installment we learned what isotopes are, and that the decay of radioisotopes is what produces radiation.
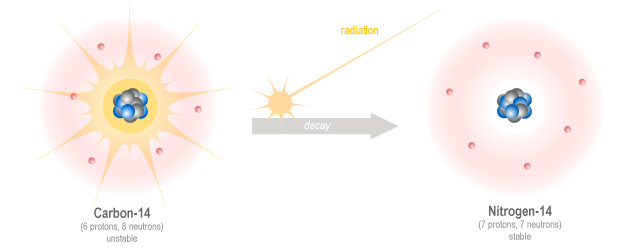
Let’s continue with our carbon example. Remember that carbon, like many elements, has a number of isotopes with different numbers of neutrons. Carbon-12 and carbon-13 are stable and are very unlikely to change, but carbon-14 is a radioisotope, which means it is unstable and will decay to become something more stable, in this case nitrogen-14. In the process it emits a type of radiation called beta radiation. Other types of radiation are alpha, and gamma. We will learn more about those later.
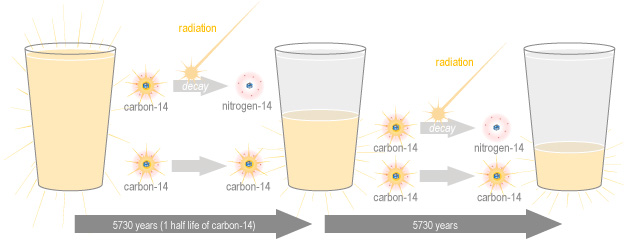
Carbon-14 decay happens very slowly, in fact it takes about 5,730 years for half of any amount of carbon-14 to decay to nitrogen-14. This is called the half-life, and is used to talk about the speed of decay of isotopes. But don’t be mistaken into thinking that an amount of isotope will completely decay in double it’s half-life, it doesn’t work that way. If you have a cup of carbon-14 and you wait 5,730 years, you will now have half a cup of nitrogen-14 and half a cup of carbon-14. If you wait another 5,730 years, half of the remaining carbon-14 will decay, so you will have three-quarters of a cup of nitrogen-14 and you will still have a quarter of a cup of carbon-14. In another 5,730 years this will reduce to an eighth of a cup of carbon-14 and so on.
OK, let’s take a look at an element we hear a lot about lately, iodine. According to the news, iodine sounds very dangerous, but actually it is essential for the healthy function of a very important gland in our neck called the thyroid.
The dangerous iodine on the news is a radioisotope called iodine-131. Stable, safe iodine has 53 protons and 74 neutrons, but iodine-131 has 53 protons and 78 neutrons. It is unstable and will decay to xenon-131 releasing gamma and beta radiation.
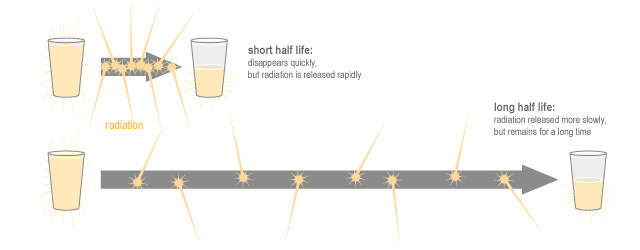
It does this quite quickly it’s half life is just eight days. This short half-life means iodine-131 disappears quite quickly, which is good, but it also means that it emits a lot of radiation in a short period of time. So half life tells us how long a radioisotope will be around, but is not a measure of how safe or dangerous it is.
Iodine-131 is of particular concern because our body cannot discern it from regular iodine, so it can accumulate in the thyroid. Lots of iodine-131 in the thyroid emitting lots of radiation can lead to cancer. But we need to ingest a lot of iodine-131 for this to happen.
In the next instalment we will look at different types of radiation.