Understanding Radiation: Radiation types

Related Articles
Following the recent problems at the Fukushima No.1 Nuclear Power Plant, the news has been teeming with talk of isotopes, radiation, contamination, and health risks. For many of us, all this talk of half lives and microSieverts can be a little confusing. In order to understand what we are being told in the news, we need to know a little about nuclear physics. The following series of articles will introduce a few fundamental concepts to help us better understand recent events (don’t worry, we’re going to keep this simple).
Previously we learned about isotopes, their decay, and what is meant by half life. Next we will look types of radiation.
Strictly speaking, radiation includes lots of harmless things like radio waves and even light. But some types of radiation have enough energy to break chemical bonds, and since there are many important chemical bonds in our body, this makes them dangerous. These types of radiation are called ionising radiation, and a very common example is ultraviolet radiation. When we hear about radiation on the news, they are talking about ionising radiation.
While any type of ionising radiation can be dangerous, some types are more dangerous in certain situations than others. This may sound a little complicated, but it is important to take a closer look at radiation types, to understand how they may be affecting us. Three main types of ionising radiation are alpha particles, beta particles, and gamma radiation.
First of all, let’s look at alpha particles.
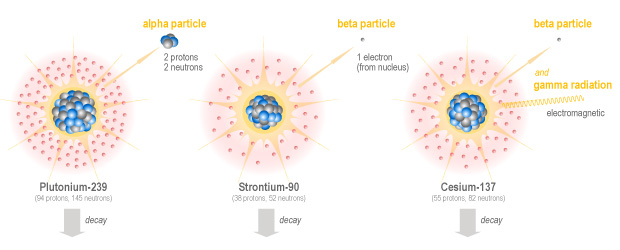
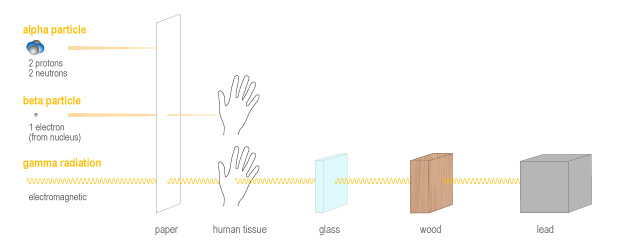
Alpha particles are made up of two protons and two neutrons. They are heavier and slower than other types of radiation, and they are highly ionizing (which means they are very destructive). Because they are heavy, slow and highly reactive, they cannot travel very far just a few centimetres in air or less than a tenth of a millimetre of living tissue. This means they can easily be stopped by a sheet of paper, or the dead cells that form the outer layer of our skin. However, although they cannot travel far, alpha particles can do a lot more damage over that short distance than other types of radiation.
So the low penetration of alpha particles means that isotopes which emit them are not particularly dangerous as long as they are outside the body. However if they are ingested (eaten or inhaled) then they can move very close to our cells, or even inside of them, allowing them to do a great deal of damage.
The next type of radiation is called beta particle. A beta particle is essentially a fast-moving electron which is emitted from the nucleus of an atom. This happens when a neutron becomes a proton during what is called beta decay. Beta particles are very fast and can carry a lot of energy (although there is a lot of variation in energy level). Beta particles are less ionizing than alpha particles, which means they do less damage, however they are able to penetrate material more deeply than alpha particles. Beta particles can travel meters in air, or millimetres in skin or living tissue. So it is possible to receive damage to the skin from nearby isotopes which emit beta particles. Of course, if these isotopes are ingested they can then do damage to internal organs.
Finally, the last main type of radiation is gamma radiation. Unlike alpha and beta radiation, which are particles, gamma radiation is a form of electromagnetic radiation, which also includes radio waves, microwaves, and visible light. Gamma radiation is very similar to x-rays. It has very high energy and can easily pass through plastic, flesh, and even steel. In fact thick sheets of lead must be used to shield from gamma radiation. For this reason, radioisotopes which emit gamma radiation can damage our internal tissues, even if they are not ingested.
So in a nutshell, an isotope which emits alpha particles is not a serious health hazard unless it enters the body, at which point it becomes very serious. Isotopes which emit beta or gamma radiation can be a problem externally if it is close and if there is a lot of it, and this also becomes more serious if it enters the body.
Taking some examples from the news, iodine-131 and cesium-137 emit beta particles and gamma radiation, strontium-90 emits beta particles only, while plutonium-238 emits alpha particles.
In the last instalment of this series, we’ll learn about the units used to measure radiation and what they mean.